Boudouard equilibrium
Describes the relationships between the reaction gases carbon monoxide (CO) and carbon dioxide (CO2) generated during carbon combustion according to the following
reaction equation (eq. 1):
Eq. 1:
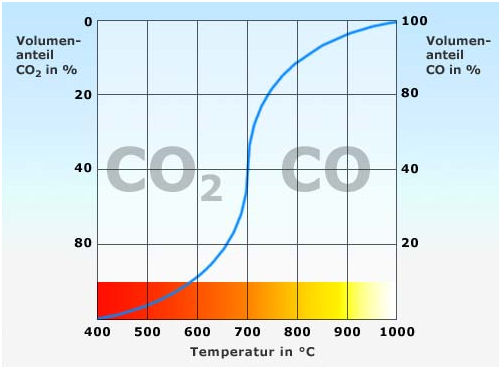
Therefore, the Boudouard equilibrium (named after Octave Leopold Boudouard, 1872 to 1923) defines a temperature-dependent equilibrium position in the combustion of carbon, e.g. charcoal as a molten bath cover or foundry coke in cupolas. Fig. 1 illustrates the temperature-dependent equilibrium position as a curve.At a low temperature, the reaction occurs from left to right, i.e. the gas mixture consists almost entirely of CO2 and the CO fraction is only small. At high temperatures of 1000°C and more, the equilibrium shifts to the left side of the reaction equation, i.e. the combustion gas only consists of carbon monoxide.
At a constant temperature, a gas mixture left of the curve in Fig. 1 continues to break down into CO2 + C until the equilibrium is reached; vice versa, at the same temperature, the CO2 in a gas mixture on the right side of the equilibrium curve will continue to react with the carbon also until the equilibrium is reached. Reductions in temperature and pressure automatically result in a change in the equilibrium state.