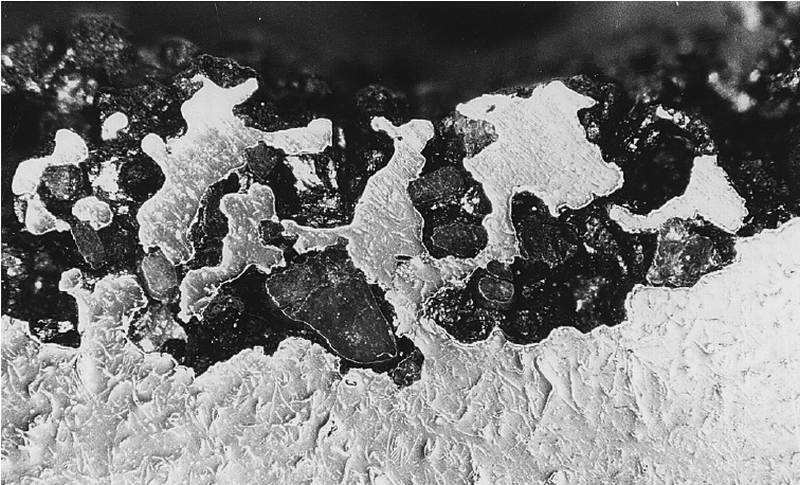
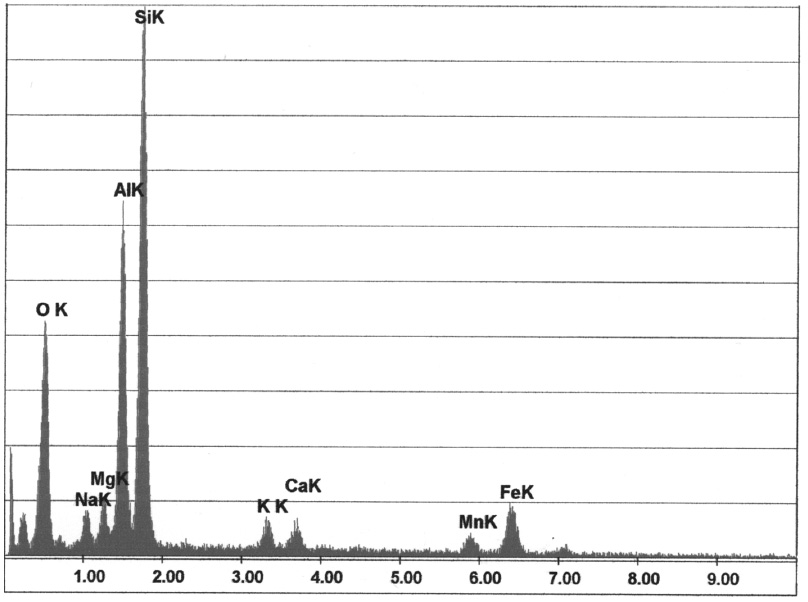
Fayalite
Fayalite is an iron silicate of the composition 2FeO·SiO2 which is present in the bentonite-bonded mold material system and reduces the melting point.
At high temperatures (casting temperatures of cast iron and steel), both the quartz sand and the decay products of clay react with metal oxides (e.g. FeO) to form low-melting orthosilicates according to the following reaction scheme (eq. 1 and 2):
Eq. 1:
Eq. 2:
The resulting iron oxides wet the mold material much better than metallic iron, allowing for increased sintering and burning effects (see Burnt-on sand). Fig. 1 shows pronounced sintering of a casting, Fig. 2 shows the corresponding element spectrum from an EDX analysis.
In general, it can be expected that reactions between metal and mold material result in intermediate layer phases which show a different behavior regarding wettabiliy and reactivity depending on the reactants’ composition.Such reactions with metal oxides can be prevented or reduced using chemically neutral basic mold materials. The more intense form of burning or sintering is metal penetration. The physicochemical reactions are then amplified by previous penetration of metals or metal vapors.