Foaming
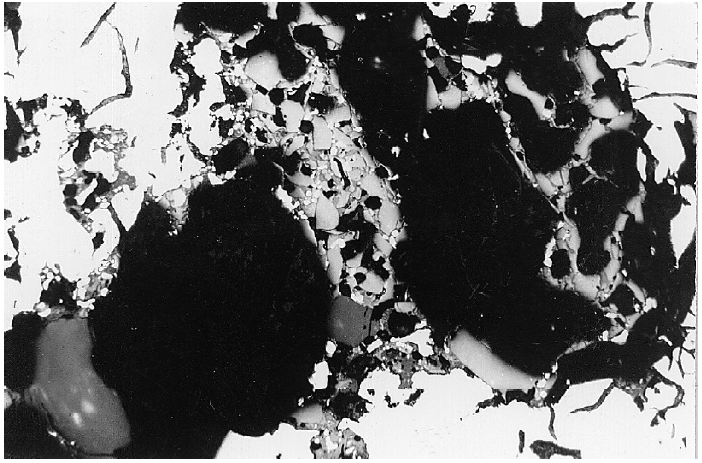
Non-metal inclusions which can be classified as slag according to their composition (Figs. 1 and 2).
These inclusions can have a round or streak-like shape. Manganese sulfide inclusions frequently occur in the area where they appear and micro-shrinkages or gas pores are very frequent in the vicinity of these non-metal inclusions.
When compared to unaffected areas of the structure, chemical averaging analyses show accumulations of manganese, silicon and sulfur which can exhibit an appearance similar to slag inclusions. In extreme cases, small slag spots are formed at the casting surface which leave a pitted surface upon removal (see also White film).
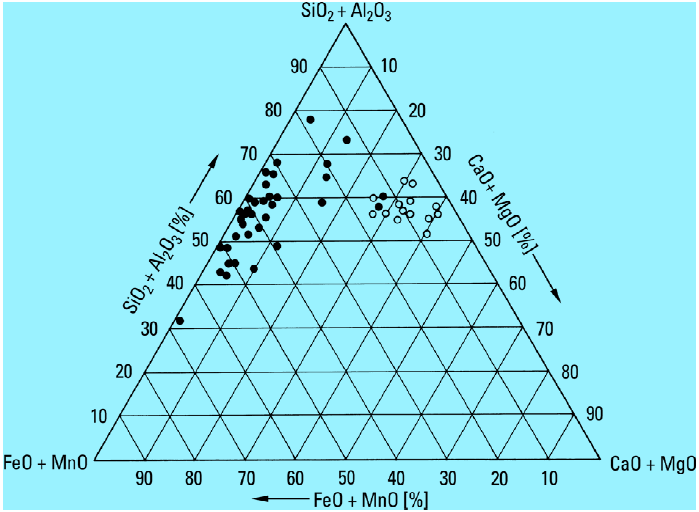
Foaming is caused by ladle slag and not by furnace slag as often suspected and assumed.This can easily be seen when looking at the position of the two slag sections in Fig. 3 for the reduced system (FeO + Fe2O3 + MnO) – (SiO2 + Al2O3) – (CaO + MgO). Since the metal oxide fraction in copula slag is intended to be as low as possible as every percentage point of metal oxides in the slag is tantamount to an oxidation-induced loss in alloying elements in the melt, copula slag is chemically composed of higher fractions of SiO2/Al2O3 and CaO (right side of the concentration triangle in Fig. 1) and lower fractions of FeO/Fe2O3 and MnO.
The fact that the ladle slag and the analyzed foam are similar in chemical composition and that the different crystal types show conformity in radiographic images indicates that foaming and slag formation in the ladle are caused by the same processes.
Furthermore, it should be noted that the melting point of ladle slag is relatively low at higher manganese oxide levels. The eutectic in the system 2Fe SiO2 – MnS lies at about 10% MnS and has a melting point of 1050 to 1060°C. This means that the slag is still liquid during solidification of the iron. The gas blisters found are likely formed by a reaction between the iron oxide fraction in the slag and the graphite precipitating during solidification of the iron according to FeO + C → Fe + CO; the resulting gas blisters can no longer escape and are captured in the area of the slag zones, creating the typical foaming image.
The ladle slag itself is formed by an interaction between iron and atmospheric oxygen (oxide film formation) and not so much or not at all by segregations of oxides previously dissolved or suspended in the iron. In this context, the chemical composition of the iron is of great importance for the temperature at which the oxide film is formed during cooling and for its physical properties and chemical composition. As an example, the oxide film formation temperature is increased by silicon and reduced by manganese. In addition, the sulfur content also has a great influence on this temperature; 0.1% S increases it by about 100°C.
Melts containing excessive amounts of titanium and aluminum, i.e. elements with high oxygen affinity, also show a tendency towards oxide film formation. The length of the interval at the end of which the bath is completely covered with slag at a decreasing temperature isalso differs within a wide range; manganese extends this interval.
Therefore, the aim is to create a melt with a low oxide film formation temperature in order to achieve the highest possible range of operation between the oxide formation temperature and the casting temperature so that the melt can be cast at a temperature at which ladle slag formation is still low. Ladle slag can reach the mold cavity during casting or even form there.