Hydrogen solubility
Hydrogen solubility rapidly decreases from approx 1 cm3/100 g at casting temperature in liquid aluminum to approx. 0.05 cm3/100 g in solid metal at solidification temperature (see Solidus temperature) and by the factor 20, see Fig. 1.Hydrogen is mainly atomically absorbed over the melt surface and by means of dissociation of the water molecules in the air (air with 65 % relative humidity at 25 °C contains 16 g/m3 H2O vapor!) according to reaction equation 1:
Eq. 1:
The oversaturated, atomically dissolved hydrogen in the aluminum melt can be recombined to H2 molecules and diffuse as blisters (Hydrogen porosity) if the surface energy for generation of the blister surface is overcome.
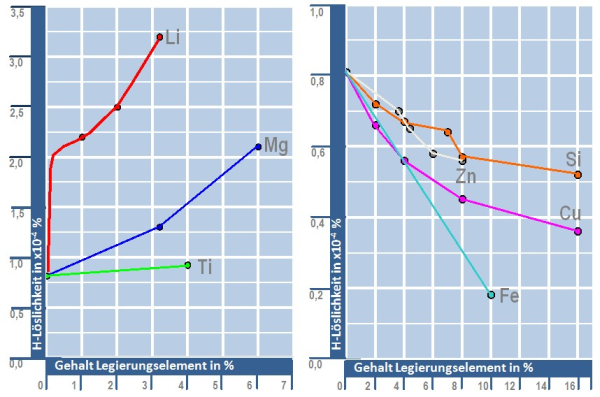
The volume of the hydrogen dissolved in the aluminum melt depends on the partial pressure of the hydrogen in the atmosphere, the composition of the alloy (Fig. 2) and the melt temperature according to the following equation 2:
Eq. 2:
The hydrogen content of the melt can be determined by direct measurement by applying the Chapel process, for example.For this reason, the formation of pores occurred mainly at already existing interior surfaces, e.g. at the boundary surfaces between metal matrix and oxidic contamination or precipitations during the solidification process. The reduction of the hydrogen content in the meld and the content of non-metallic contamination is an important factor for the quality of the casting. Similarly, it is also necessary during the casting process itself to prevent additional absorption of hydrogen and gas which may be caused by humidity in the molding material or the coating at the mold surface.
Hydrogen sources include (according to R. Klos):
- Atmosphere (it contains water vapor)
- Humidity from fireclay / furnace lining
- Humidity from tools
- Corroded charge materials, e.g. ingots and scrap after the reaction Al(OH)3(s) + Al(l) → Al2O3(s) + 3H
- Oily circulation materials, chips or scrap
- Salts, treatment agents
- Combustion gases
Generally, the oxide film or scum on the melt surface protects the aluminum from excessive absorption of hydrogen. Refined and overheated melts increasingly tend towards hydrogen absorption. If casting parts are to be welded together, the atomically dissolved hydrogen also diffuses in form of gas bubbles during remelting. At higher concentrations, this also leads to gas formation and swelling of the weld seam (relevant for vacuum-assisted die casting, see alsoVacuum systems and vacuum valves from VDS SA Vacuum Diecasting Service or for weldable and heat-treatable die cast parts).
Additional references:
Hydrogen determination
Rotary degassing