Low-alloyed ductile iron
The mechanical properties of ductile iron defined in standards and regulations is based on evaluations of unalloyed iron without any heat treatment, except thick walled castings of the grades GJS-700 and GJS-800 (the Pearlitic matrix can just be achieved by alloying elements). Therefore it is a relative unknown fact that alloying achieves substantially more favorable combinations of strength, toughness and, above all, of the 0.2% offsetyield strength. This can be applied as-cast, as well as heat treated. Besides this, the toughness of such cast iron does not need to decrease in the Pearlitic state, while alloyed ferritic iron with optimized compositions can achieve good values of impact strength. The suitability of the cast iron for certain applications can be improved by the addition of alloying elements, i.e. by the desired influencing of the matrix and its hardness. This presupposes that the nodularity of the graphite, the nodule count and the content of non-metallic inclusions meet the requirements.
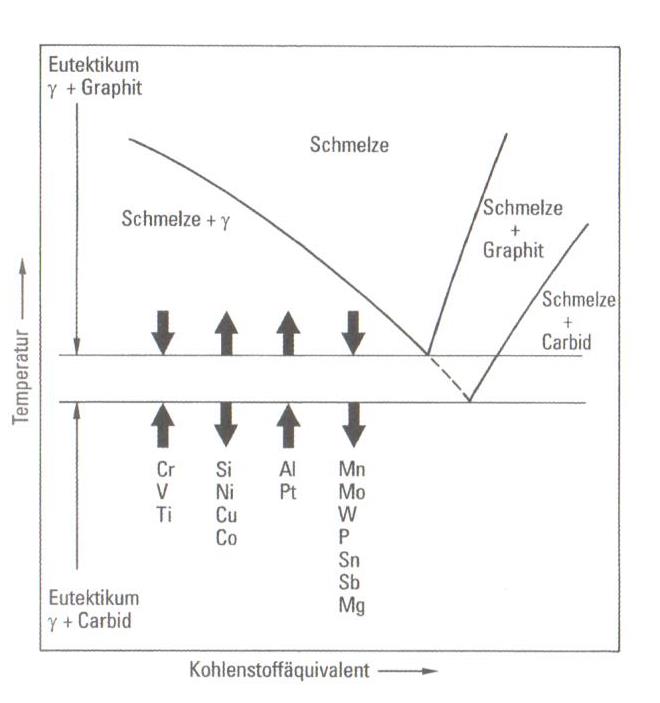
Alloying elements essentially influence the chill tendency and carbide formation. It has to be mentioned first that most alloying elements shift the eutectic temperature of the stable iron-graphite system and consequently obtain a change of the carbon equivalent which directly influences the chill. In addition, the alloying elements change the equilibrium solidus temperature of the iron-graphite-eutecticum (stable system) and of the iron-carbide-eutecticum (metastable system) as given in fig 1.
Because the alloying elements influence both eutectic temperatures, they affect the range of the interval between both eutectic temperatures and subsequently the supercooling intensity which is still admissible for chill avoidance. With regard to the chill the effect of the alloying elements is equal to or greater than the effect on the carbon equivalent.
The effect of alloying elements on ductile iron properties is based on its influence on the ferrite/pearlite ratio of the matrix and on the work-hardening of ferrite and pearlite. Alloying elements increase the ferrite strength, above all by solid solution strengthening, as well as the hardness of the pearlite by decreasing the lamellar spacing between cementite and ferrite. The ferrite/pearlite ratio is influenced by various processes; among others the influence on the eutectic transformation temperature and the transformation kinetics.
The extent of work-hardening by alloying elements depends upon the matrix formation, based on the different work-hardening processes of ferrite and pearlite. The extent of the work-hardening of an iron matrix, consisting of ferrite and pearlite, depends upon the ferrite/pearlite ratio. By adding silicon for example to a Pearlitic iron the strength decreases, because of the increasing ferrite content of the matrix. In case of a ferritic iron increasing strength can be observed by solid solution hardening of the ferrite.
The addition of copper and manganese yields a considerable increase of strength for ferritic iron. In addition to the solid solution hardening of the ferrite and pearlite both increase the pearlite content of the matrix. For cast iron, already having a Pearlitic matrix, increasing manganese and copper content result in increased strength by the work-hardening of the pearlite, but the gradient is flatter than that of the Pearlitic/Ferritic structures. Charting the strength against the content of alloying elements in a diagram, two sections are obvious: one section with steep rising strength by increasing content of alloying elements and a second section with a more flat gradient (fig. 2). The transition from the first to the second section is the point at which a total Pearlitic matrix exists.
Additional references:
Ferrite
Cementite
Structure
Structural matrix
Structure formation of cast iron
Metal matrix of cast iron
Maurer diagram