Metal matrix of cast iron
Metal matrix of a cast iron alloy that depends on the composition of the alloy and the solidification conditions.
Control and formation of the matrix is based on the uniqueness of the dual iron-carbon system (s. Iron-carbon phase diagram), which has made ferrous metals the most important metal materials by far. Characteristics are:
- Transformation of the face-centered cubic austenite lattice into the body-centered cubic ferrite lattice, and
- Insolubility of carbon in ferrite, resulting in carbon precipitation and thus enabling precipitation of carbon both in the form of graphite and in the form of iron carbide (cementite).
Therefore, it is generally possible to obtain five different transformation products and the corresponding matrices for cast iron:
1. Ferrite, with the transformation of austenite taking place in the stable system. In microscopic scale of the structure, this represents long-term diffusion. It takes relatively long and/or requires comparatively high temperature levels. With a ferritic matrix (Fig. 1), maximum toughness, thermal resistance and good machining properties can be obtained.
2. Pearlite, in which carbon is precipitated as cementite in the form lamellae (Fig. 3) along with ferrite (Fig. 2). The transformation to pearlite in the meta-stable system corresponds to medium-term diffusion of carbon. The distance between the lamellae decreases as temperature falls.
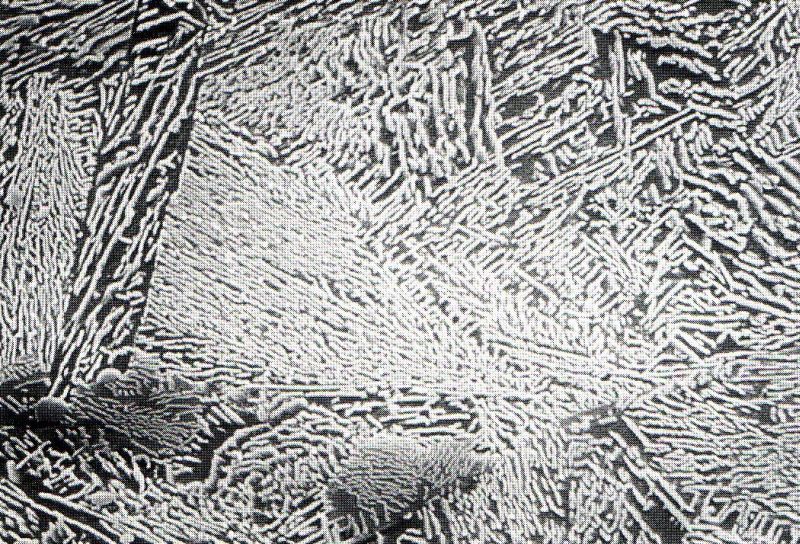
3. Ausferrite, below a temperature of 400 to 500°C, the type of transformation mentioned above is no longer possible; instead, short-term diffusion takes place which initially only generates ferrite. The carbon displaced from the ferrite accumulates in the austenite, whose carbon content may increase up to 2 % due to this process. This stabilizes the austenite down to low temperatures and the resulting structure comprises a mixture of finely acicular ferrite and austenite. This is the structure aimed for in ADI (see Bainitic cast iron) (Fig. 4). A critical prerequisite for successful performance of these measures for improvement of strength and toughness combinations is undisturbed formation of the solidification structure.
4. Bainite, forming upon longer hold times in the temperature range between 400 and 240°C since the carbon-rich austenite in ausferrite segregates to form ferrite and acicular carbide so that bainite (known from steel production) is obtained Fig. 5). This structure is unwanted in ADI.
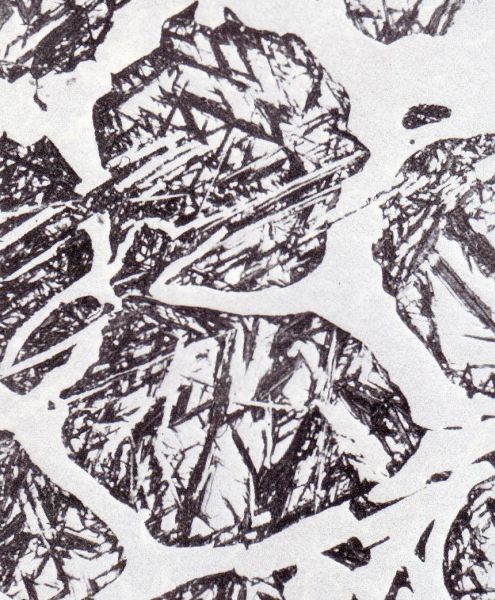
5. Martensite, formed by diffusion-free transformation of austenite. The precipitation of carbides is suppressed and the α-solid solution supersaturated with carbon is distorted tetragonally (Fig. 6). Below a temperature level of approx. 240 °C carbon diffusion is not longer possible, the austenite lattice collapses without diffusion. These transformation processes of austenite can be strategically influenced by heat treatment and/or alloying elements.