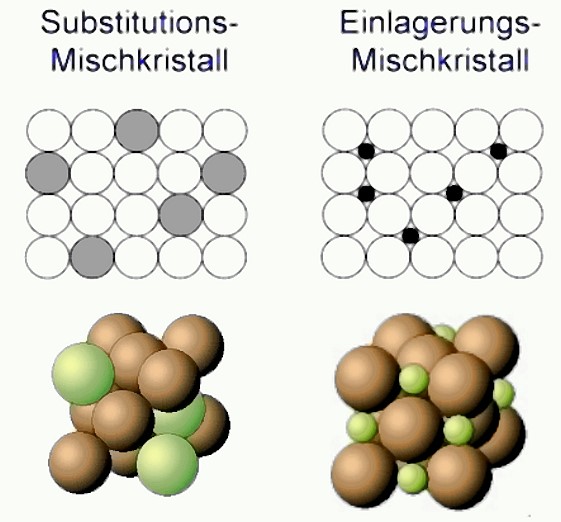
Mixed crystal
A “solid solution” (“ss”) refers to a crystal or crystallite consisting of at least two different chemical elements, with the foreign atoms being statistically distributed within the host lattice.
However, what is characteristic about a solid solution is that the lattice structure of the initial crystal remains unchanged by the integration of a second particle type, which must have a similar crystal lattice. Thus, the original atomic lattice of an element is conserved, whether the foreign atoms are integrated into the host lattice interstitially or substitutionally.
Most metals are able to incorporate other atom species into their atomic lattice structure (matrix). These foreign atoms can either be integrated at interstitial sites of the lattice or occupy a site in the atomic lattice of the matrix. Accordingly, the following types of solid solution are distinguished (Fig. 1):
- Interstitial solid solutions
- Substitutional solid solutions
If they exhibit metallic properties, solid solutions are also known as alloys.
Due to the different lattice parameters, the lattice is always more or less strained, resulting in an increase in strength (so-called solid solution hardening or solid solution strengthening, see also Strengthening mechanisms). Based on this, solid solutions have a higher hardness and tensile strength and a lower melting temperature than the pure material. Moreover, metallic solid solutions exhibit reduced electrical conductivity.
Examples of solid solutions formed by interstitial atoms include the Fe-C system.
Zn in Cu or Cu in Al are examples of strengthening through substitutional foreign atoms.