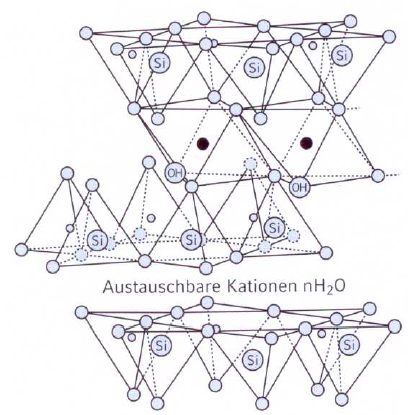
Montmorillonite
Layer silicate which has a three-layer structure (Fig. 1) consisting of two tetrahedron layers and one octahedron layer (s. Three-layer mineral).
The layers are not rigidly connected and can swell as a result of the storage of water. Montmorillonite is the major component (60-80%) of bentonite. The water molecules are bonded to the clay minerals by their surface activity and develop a water shell surrounding the particles. The swelling of the bonding clays is largely influenced by the ion deposits and their packing density (ion exchange capacities). Bentonites with Na+ ion deposits can virtually swell without limit, whereas the swelling capacity of Ca++ bentonites is limited due to the lower water water binding capacity.
Additional references:
Smectites, Illite
Calcium bentonite
Sodium bentonite