Peritectic
In alloy systems with complete solubility in liquid state and complete insolubility in solid state. A peritectic system is characterized by the fact that a solid phase β with the concentration cβ at the peritectic temperature Tp melts under decomposition.
During the peritectic reaction, there is a liquid phase S and a solid phase α in thermodynamic equilibrium with a solid phase β whose chemical phase is different to that of phase α. The peritectic is therefore a kind of separation (s. Peritectic line).
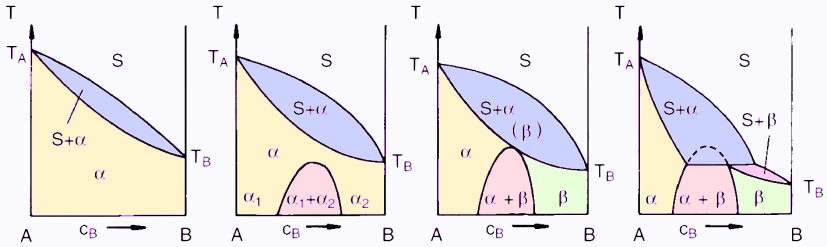
The development of a peritectic system starting from a system of complete miscibility through the increasing formation of a miscibility gap is depicted in Figure 1.
Figure 1: Schematic development of a peritectic system with an increasing miscibility gap in solids
The peritectic reaction happens as follows: S+ α -> β
As the temperature sinks, α crystals form which react with the melt at a constant temperature, forming β crystals. The β crystals deposit around the α crystals, hence the name peritectic (peri = around). Peritectic systems generally occur when the melting points of the pure components are very different. The peritectic temperature then lies between the melting points of the pure components. The Pt-Re system is given as an example.
Additional references:
Peritectic line, Peritectoid
Eutectic, Eutectoid