Segregation
Formation of microscopic and macroscopic differences in composition in an otherwise homogenously structured alloy that consists of mixed phases.
The solid phase that solidified first has a different alloy content as the residual melt. Accompanying and alloy elements considerably influence the segregation behavior. For example, this may lead to inclusions, carbide or structural degeneration at the grain boundaries influencing the mechanic and particularly the dynamic properties of the material.
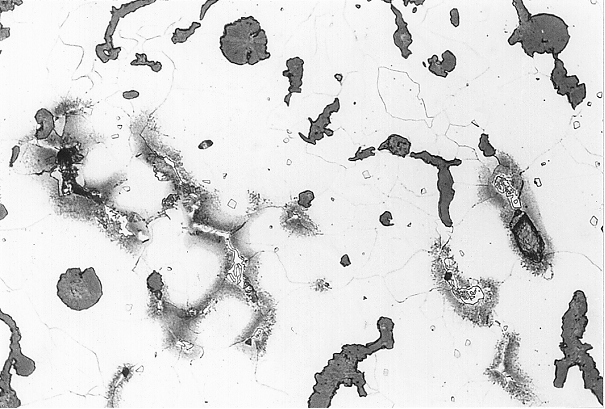
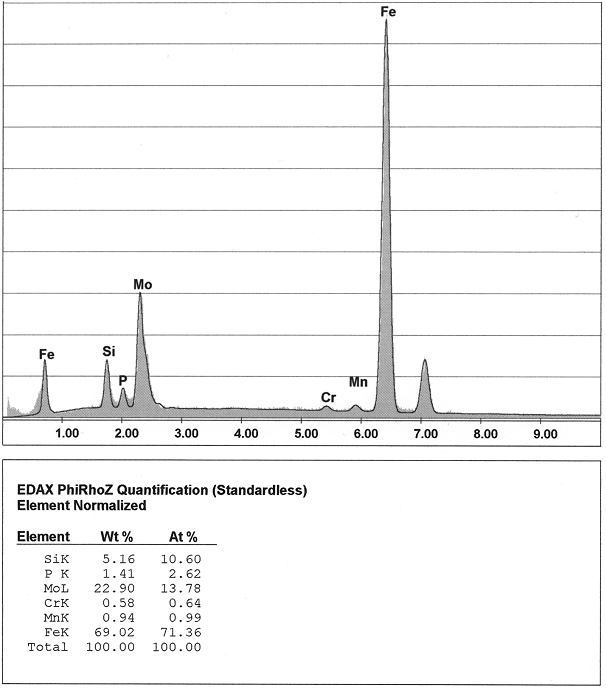
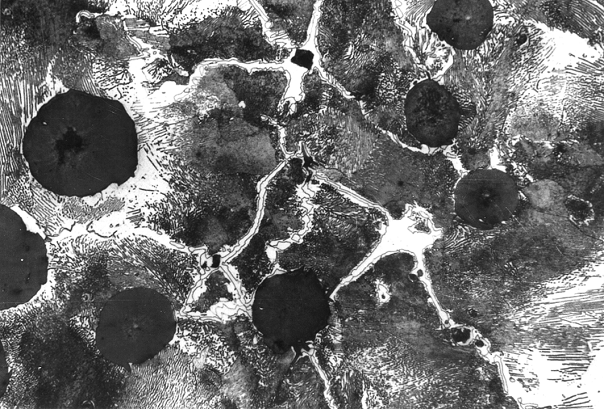
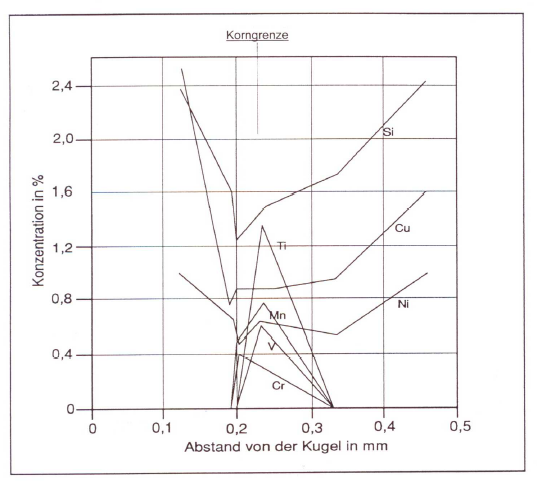
These degenerations can be clearly identified at the metallographic specimen (Fig. 1). Direct determination of the anomalies are not possible by means of metallographic examination. Examinations with the an electronic micro probe, however, lead to clear determination of the degenerated or enriched elements at the grain boundaries (figures 2 and 3).
Generally, a differentiation is made between macro segregation and micro segregation.
Gravitational segregation, ingot segregation and reverse ingot segregation are macro segregations with limited local decomposition, i.e. differences in composition in the solidified casting with respect to the total melt.
In contrast to this, micro segregation (crystal segregation) refers to decomposition of mixed crystals, i.e. in case of differences in the composition on a microscopic scale.
Potential causes of gravitational segregation include considerable differences in density between solidified primary crystals and the residual melt. The specifically heavy crystals sink in the melt and accumulate at the bottom or in the lower range of the casting. Specifically lighter components rise in the melt and accumulate at the surface of the bath or in the upper range of the casting.
This way, segregation ranges with accumulations of primary silicon crystals (primary silicon) easily form in hypereutectic AlSi alloys. This applies particularly, if the primary crystals have a rough structure. Known are also gravitational segregations of primary graphite in hypereutectic nodular graphite cast iron, which rise in the melt as graphite flotation or as kish graphite in hypereutectic gray cast iron.
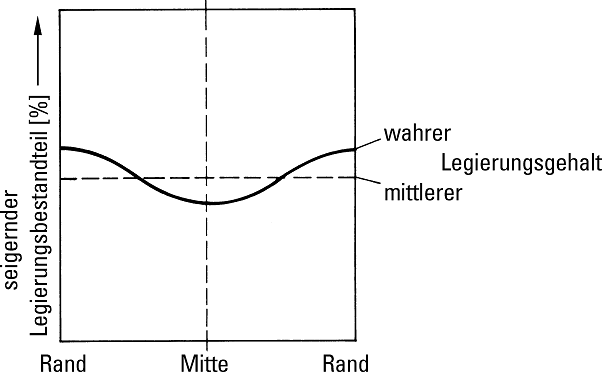
Ingot segregation is caused in cast ingots or thick-walled castings by decomposition or concentration differences in the liquid residual melt if no flow is provided to balance the concentration with the already formed crystals. The proportion of the related alloy component subject to segregation is lower at the edge of the casting and higher in its thermal centre (Fig. 4).
The residual melt enriched with a specific alloy element due to lack of concentration equalization is displaced towards the outside of the casting due to solidification pressure or the suction effect in the capillary cavities between the solidified crystals. As illustrated in Fig. 5, the alloy content in the edge areas is higher than in the center, which can be regarded as the formation mechanism of reverse ingot segregation.
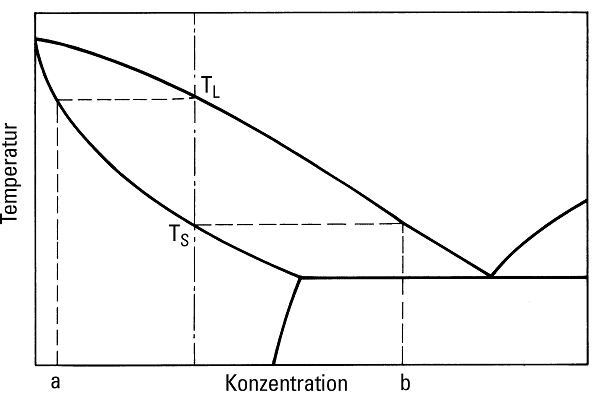
As illustrated in Fig. 6, primary crystals of composition (a) are diffused in the beginning of the solidification of a mixed crystal-forming alloy after the temperature has fallen below the liquidus temperature TL while the residual melt that solidified last complies with concentration (b) when reaching the solidus temperature TS. These differences in concentration between primary crystals and the residual melt that solidified last are usually compensated by diffusion in such a way that the structure consists of uniform mixed crystals with the same composition. If this is not the chase, defects occur during concentration equalization, i.e. segregation at the edges of the mixed crystals. This micro or grain boundary segregation is higher the lower the diffusion tendency and the wider the solidification interval (figures 7 and 8).
For example, the segregation in slowly solidifying castings from nodular graphite cast iron, has a strong influence on the solidification structure and on the mechanical properties to be expected. The segregation behavior of the alloy elements and the trace elements is different.
According to their segregation behavior, the elements can be distinguished in two groups: indirectly segregating elements like silicon, nickel, copper, etc., which are accumulating in the first solidifying iron (primary austenite or austenite shell around graphite spherolites) as well as in directly segregating elements like manganese, chromium, vanadium, titanium and phosphor occurring in the iron solidifying last (residual melt). The slower a casting is solidifying, the larger are its eutectic grains (see Eutectic grain count) and the more distinctive are the concentration differences at the eutectic grain boundaries. These grain boundary segregations can lead to carbide diffusions (e.g. in case of silicon, chromium or maganese segregation) or graphite degeneration (e.g. in case of titaniumsegregation or magnesium depositions) (Figures 9 and 10).
Silicon increases the yield strength and the tensile strength of the metal matrix, has an increasingly embrittling effect and stabilizes the formation of ferrite. In case of stress, the yield strength is first exceeded at the Si impoverished “residual melt ranges”.
The more solid mixed crystal around the graphite nodules and in the dendrites is less involved in deformation. This has the positive side effect that lesser graphite degenerations are buffered by the less deforming accumulation shell, however, the restriction of fluid processes on the grain boundary ranges leads to an inhomogeneous sliding distribution in the material and to the occurrence of internal micro-stresses. Both reduces elongation at failure.
Micro segregation of the phosphor is an additional risk. This element also accumulates in the residual melt range (see Phosphorous eutectic). Here, it stabilizes gray solidification, however, it considerably impairs the strength of the material. This is also the reason for the limited level of silicon and particularly phosphor in ferritic types with a defined impact strength (EN-GJS-400-18-LT).
Please note that, in connected segregation areas, which are the result of accumulation or impoverishment of accompanying, alloy or interfering elements, there is a high concentration of the above mentioned elements although concentrations which are generally referred to as harmless have been found in the chemical analysis. Additionally, trace elements in the residual melt ranges as most important micro segregation zones form additional sub-segregations that increase the negative impacts of impoverishment or accumulation.
The following counter actions are to be recommended:
Gravitational segregation
In this respect, a good and mostly even the only way to decrease the solidification time additional cooling (denseners, chills) in the mold as well as in the cores.
Ingot segregation
Ingot segregation often occurs in aluminum and copper alloys (e.g. tin bronze, red bronze, phosphor bronze, tin lead bronze) and particularly in continuous cast and ingot material, however also in real castings, and can be mostly avoided by the following measures:
- Correct melting, particularly with largely applied degassing of the melt with oxidizing pretreatment,
- Avoidance of unnecessary superheating temperatures and inadmissibly high casting temperatures. For AlSi alloys, this also applies for already grain refined melts
- Reduction of the holding times
Micro segregation
The effective measures to reduce micro segregation include:
- Checking of charge materials for accompanying and trace elements
- Suitability test of the treatment alloy (sulfur and phosphor content, content ofrare earth metals)
- Late inoculation treatment (mold or late stream inoculation) to ensure fine distribution of the nodular graphite and prevent silicon degeneration at the grain boundaries
- Prevention of oxygen absorption, for example due to humid charge materials or insufficiently dried ladles, mold and casting-related measures to reduce the solidification time and to ensure a cooling rate that is as even as possible.
If segregation occurred which lead to the formation of boundary grain carbides, for example, there is the possibility to partly reverse this segregation by annealing treatment (diffusion compensation).