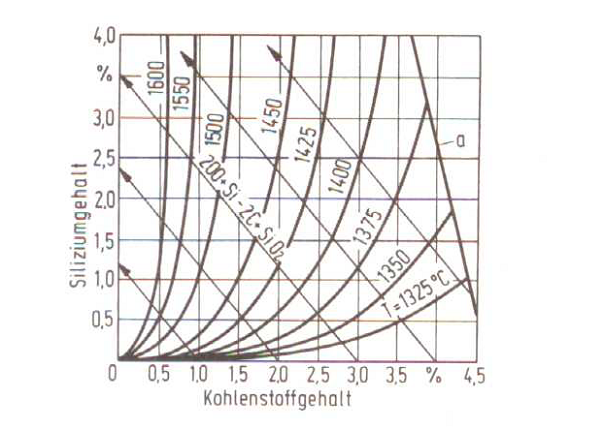
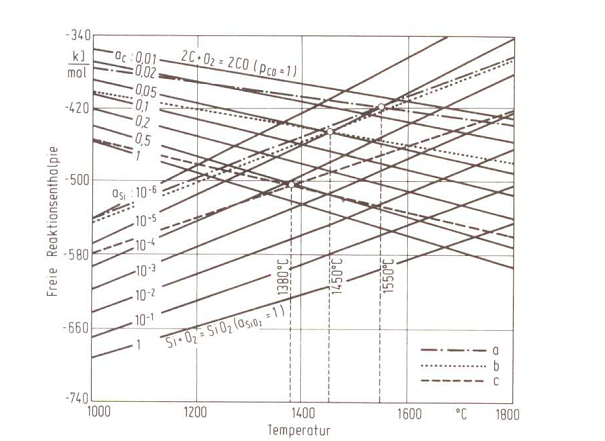
Equilibrium temperature
Crucible reaction temperature which is reversible and where the partners of the reaction have an equilibrium. In case of iron carbon alloys and in presence of silicium and oxygen it proceeds according to the following equation (Equ. 1) :
Equ 1:
If solid silicic acid develops below the C/Si isotherm (equilibrium temperature), dross develops. The C/Si isotherms for cast iron with flake graphite with normal carbon and silicon contents of approx. 3.5 to 4.0 percent of C and approx. 1.5 to 3.0 percent of Si lie between 1400 and 1440 ° C.
Growing silicon contents make the TG increase, grwong c contents make them decrease. In this case, silicon determines the melt oxygen content through silicic acid development (auto-desoxidation with silicon) at temperatures below or at the TG, silicon burns and creates SiO2 which passes over to the slag phase.
At temperatures above the TG, carbon dominates the melt oxygen content; SiO2 is reduced under {CO} development, i.e. The amount of silicon in the melt increases. Due to delays in the phase development, the CO development sets in later, i.e. only at the so-called cooking temperature (TK). According to K. Orths, W. Weis and M. Lampic, there is the following relation between the equilibrium temperature TG and the relevant cooking temperature TK for acid-lined crucible induction furnaces (Equ. 2):
Eq. 2:
Fig. 1 shows a relation for the equilibrium temperature between the carbon and silicon content, Fig. 2 shows a free reaction enthalpy for C and Si oxidation.