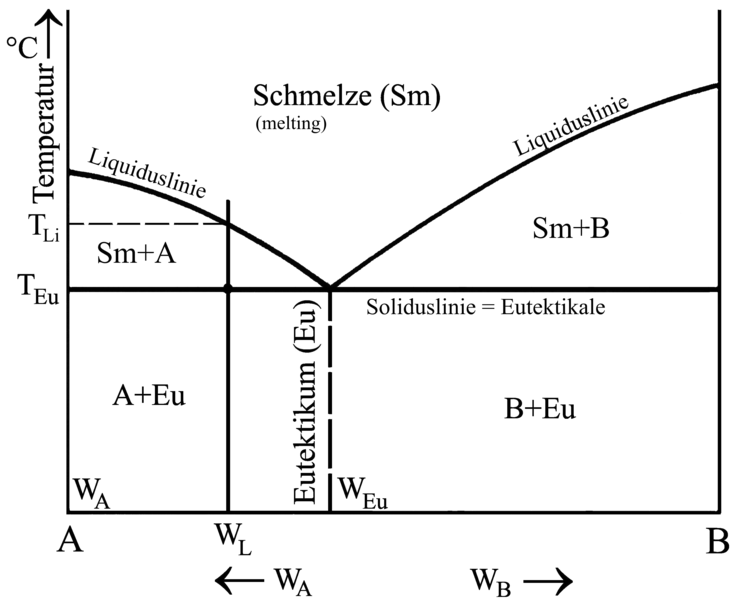
Eutectic
Refers to a mixture of two or more substances that are absolutely immiscible in solid state and can only be completely mixed with each other in molten state.
A eutectic mixture has a lower solidification point than all other mixtures containing the same constituents. Also, a eutectic does not have a solidification interval, its liquidus temperature is identical with its solidus temperature (Fig. 1). All alloy components that are separate in solid condition due their insolubility form the structure of the eutectic. The eutectic is always present with very fine grain distribution.
Hypoeutectic alloys (alloys located on the left of eutectic composition region) diffuse A primary crystals plus eutectid during solidification; alloys located on the right of that region are hypereutectic alloys and diffuse B primary crystals plus eutectic. Nearly eutectic alloys are materials whose composition is very similyr to that of eutectics.
Additional references:
Solidfication type
Substitutional solid-solution
Solid-solution