Intermetallic phase
An intermetallic phase (also intermetallic compound) is a chemical compound consisting of two or more metals.
In contrast to alloys, the intermetallic phase shows a lattice structure which is different from those of the component metals (so-called superstructures). The lattice bond of the various atomic species is a mixture of a predominantly metallic bond and lower fractions of other kinds of bonds (covalent bond, ionic bond), conferring special physical and mechanical properties to this phase.
Very often, intermetallic compounds are hard, i.e. they exhibit high hardness, brittleness and strength and usually have a relatively high chemical resistance (good corrosion resistance). Moreover, they are characterized by a high melting point and high electrical conductance which is generally one order of magnitude greater than that of pure transition metals.
There are intermetallic compounds with stoichiometric compositions according to the usual metal valencies and there are intermetallic phases having more or less extensive ranges of homogeneity in the phase diagram. A homogeneity range, also referred to as phase range, defines boundaries within which the quantity ratio of the individual metals may vary.
The most important intermetallic phases are:
- Laves phases (type AB2, e.g. CaMg2)
- Heusler phases (ABC, A2BC, e.g. Cu2MnAl)
- Zintl phases (type AB, between alkaline and alkaline-earth metals and electronegative elements of the 13th to 16th main group, e.g. NaSi)
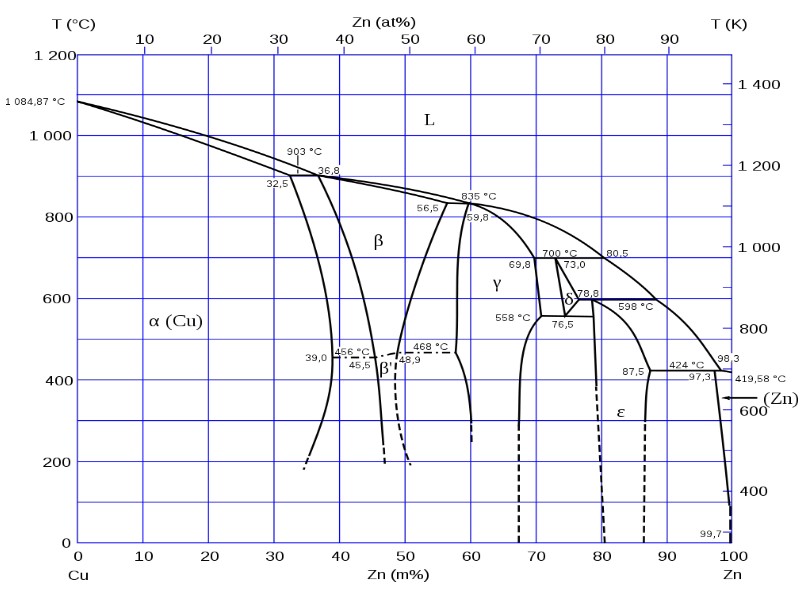
- Hum-Rothery phases, characterized by their fixed valency-electron concentration (α-, β-, ψ-, δ-brass, see Fig. 1)
High-alloy bronze and also brass alloys are composed of intermetallic phases which can be present in different forms depending on the mixing ratio (e.g. Cu3Sn and Cu3Sn5). If the correct mixing ratio is not achieved exactly, this produces classic alloys which, however, consist of the various intermetallic phases nearest to the mixing ratio.
Additional references:
Iron needles
Literature references:
Gottstein G., Physikalische Grundlagen der Materialkunde, Springer-Verlag Berlin Heidelberg, 2007.