Reduction melting
Critical metallurgical process of deriving metals from their ores that contain their oxides by reduction melting. In this process, the oxygen is removed by means of reduction (deoxidation, deoxidizer).
In a foundry-related sense, this is referred to as melt control under reduced conditions. The furnace gases contain very low levels of oxygen.
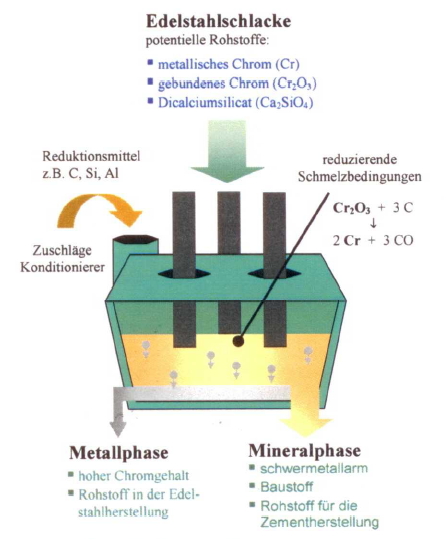
The example in figure 1 illustrates that reclamation of metals from stainless steelslag
(see Slag) is also enabled by reduction melting (Recycling of Residues from Metallurgical Industry with the Arc Furnace Technology - RecArc, EU-LIFE Environment Demonstration Project LIFE03 ENV/D/043).
In electric-arc furnaces, electrical energy is conducted through graphite electrodes into the material to be molten, in this case the stainless steelslag, to melt it.
The heavy substances in the liquid slag sink to the ground of the molten bath. There, they form a continuous phase with the lighter mineral slag cast, which still contains the chromium compounds, floating on top.
In order to also separate these bound mineral chromium contents, they have to be transformed to the metallic form of chromium. This is achieved by means of reducing agent, substances like carbon, aluminumor silicon that react with oxygen to be able to dissolve the chromium from its compounds.
As soon as the chromium compounds are reduced to elemental metallic chromium by means of these substances, it can be dissolved like the remaining metal and enters the metal phase. Reduction and metal separation take place simultaneously.
Additional references:
Oxidizing melting
Non-oxidizing melting
Oxidation
Degree of oxidation
Melting base