Flow rate in pressure die casting
The most important factor influencing the course of mold filling in pressure die casting is the rate at which melt flows into the mold. Assuming the melt behaves like an ideal liquid, the following applies to a quasi-steady flow on the basis of Bernouilli’s equation (eq. 1):
Eq. 1:
Wherein:
v = flow rate in m/s
p = flow pressure in N/m2 (Pa), with 1 bar = 105 kPa
ρl = density of the liquid alloy (kg/m3)
It should be noted that the density of the liquid alloy (ρl) is lower than that of the solidified alloy (ρS). According to an estimated approximation for aluminum, magnesium, brass and zinc alloys, density in the liquid state is about 91% of the density in the solid state at room temperature; therefore, ρl can be calculated as per
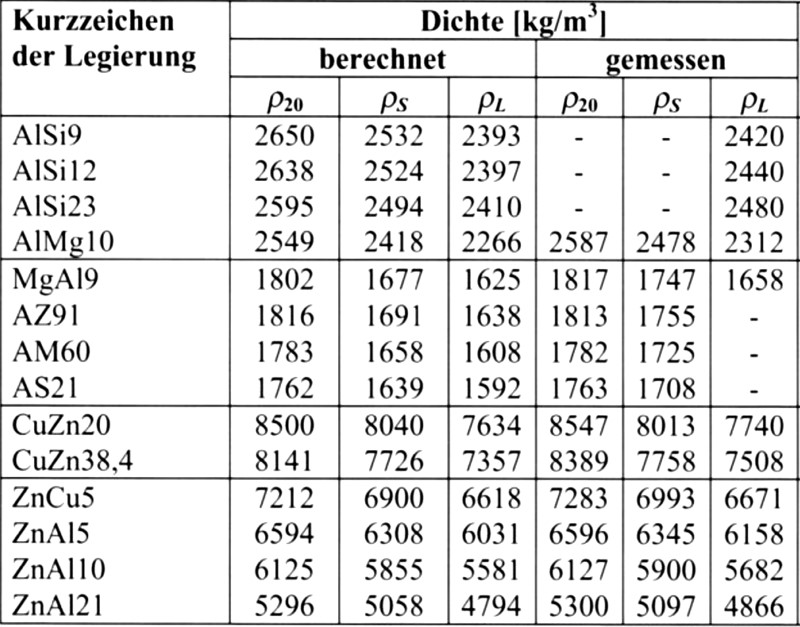
ρl = ρS · 0.91, without obtaining an overly erroneous result. B. Nogowizin performed a literature search for density values of solid- and liquid-state die casting alloys and also calculated such values from the chemical composition; these are listed in Table 1.
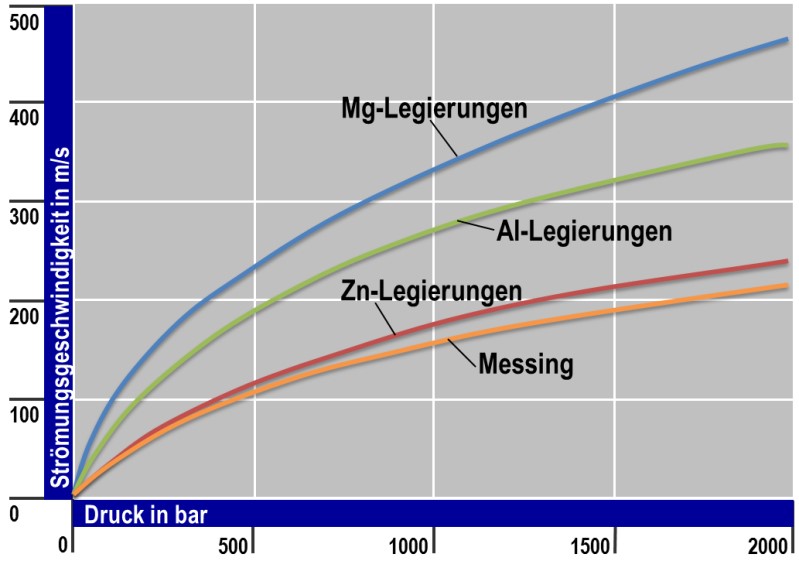
Comparing the die casting alloys based on the metals copper, zinc, aluminum and magnesium reveals a very high influence on the flow rates obtainable at the same pressure due to the different densities (Fig. 1).
For the casting materials according to E. Brunhuber, the ratio of the theoretically possible flow rates for the brass : zinc : aluminum : magnesium ratios is approx. 1.0 : 1.1 : 1.8 : 2.2.
Additional references:
Casting time diagram
Mold filling time
Mold filling time in pressure die casting
Literature references:
B. Nogowizin: Druckgusslegierungen und ihre Eigenschaften, Druckguss-Praxis (2003), no. 4, p. 161-168.
E. Brunhuber: Praxis der Druckgussfertigung, Fachverlag Schiele & Schön, Berlin 1980.