Hard spots
Inclusions of phases in the structure of casting materials of particular hardness that particularly interfere with machining and cause increase in tool wear (Figures 1 and 2).
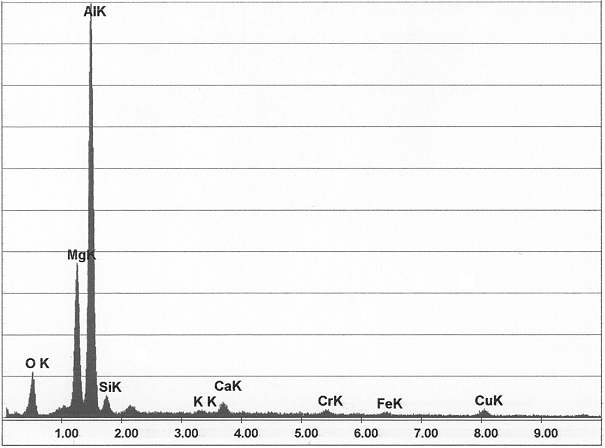
These inclusions or phases are found for example in cast iron alloys (cementite, other carbides), in cast aluminum alloys (corundum inclusions, Al-Mn-Cr- or Al-Mn-Fe segregations (see Segregation factor), Si primary crystals, s.a. Primary silicon) as well as in cast brass (Fe-Cr-Mn-B diffusions, agglomerates) and copper-aluminum alloys.
In pressure die cast parts oxidic hard inclusions are present in the form of fine, streak-like, embedded oxide skins, in the form of scattered oxide nests, as oxide skins in parallel or irregular arrangement, in magnesium-containing AlSi cast alloys in the form of spinels in “knotty” arrangement and as compact, granular inclusions of corundum type.
In cast iron alloys, hard spots manifest in the form of carbides within the structure or in the form of chill, or edge hardness at or directly underneath the casting surface. Here, the defects is clearly visible in the break structure, otherwise it is made visible in metallographic cuts. Hard spots cause weakening of the structure and along with that loss of the mechanical and dynamic properties.
The hard spots that are found in aluminum alloys (e.g. in die casting) can generally be classified in two main categories:
1. Metal inclusions (Segregation, Gravitational segregation); these spots rarely are pure inclusions but mostly intermetallic compounds with a high melting point (see Intermetallic phase) that preferably diffuse from the molten metal in iron- and mangenese-containing alloys.
The following metal inclusions are typical for castings made of Al die casting material:
Brittle crystals, originating from reactions of pure aluminum with iron (Al3Fe, Al5Fe2), crystal plates that are often cut through in a micrograph and then appear in the form of needles (β-AlSiFe5, Al9Fe2Si2, µ-Al(Fe-Si), see Iron needles), as well as intermetallic compounds of various shapes (α-AlSi(FeMn), Al15(Fe,Mn,Cr)3Si2, γ-Al(Mn-Fe)).
The causes for generation of metallic hard spots must be primarily attributed to insufficient melting or holding temperatures. Segregation of the intermetallic compounds preferably takes place at the crystallization centers. Oxides introduced by means of recirculation material or oxidized pig material serve as crystallization centers. Retarded or slow melting of this material causes prolonged duration of the “doughy phase” of the melt. The oxides or the aluminum and the alloying elements generated in this way cannot be clearly separated from the already melted metal, remain within the melt and can thus form mixed oxides or act as nuclei for segregation of intermetallic compounds.
2. Non-metal inclusions (s. a. Oxide inclusion) are available in a large variety of types and result from greatly varying causes so that in practice it is often difficult to classify them exactly and accurately, or precisely determining their origin. Frequently occurring non-metal inclusions are corundum (α-Al2O3), spinel (Al2O3.MgO, Fig. 3), silicon carbide (SiC, Figures 4 to 6) and quartz (SiO2)
These non-metal inclusions originate from oxidation of the aluminum during melting, particularly in case of excessive bath agitation and/or too high temperatures. Moreover, aluminum oxides are readily formed and introduced into the melt when excessive amounts of recirculation material are used; this particularly applies for burr scraps and overflow material with relatively large surfaces. The oxide formed first (γ-Al2O3) is relatively soft but through the heat and over time it is transformed to the very hard alpha modification (α-Al2O3), also referred to as corundum.
Oxides and oxide nests mostly originate from the molten bath. The following factors come into question with regard to formation causes:
- Insufficient purification of the melt
- Heavily contaminated and small-sized recirculation material
- Transfer of metal between vessels, heavy stirring of the bath
- Repeatedly pushing back the surface oxide skin
Additional references:
Segregation factor, Segregation factor
Gravitational segregation
Crystal segregation
Oxide inclusion
Iron needles