Peritectoid
In binary systems, the peritectoid is the transformation of two solid phases in an alloy system, forming a new phase. These reactions happen in a similar way to peritectic transformations (see Peritectic), however, they only occur in the solid state.
While, in a peritectic reaction, the melt and the phases separated from it take part, in the peritectoid transformation, only solid phases are involves: two solid phases transform into a third solid phase.
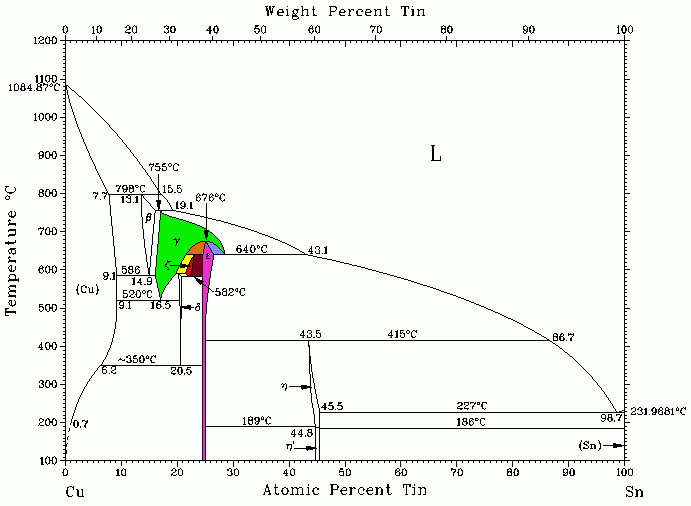
Figure 1 shows the Cu-Sn phase diagram, in which several peritectoid reactions take place.
As an example, Figure 2 shows the area between 32 and 40% by weight of Sn where a peritectoid reaction between the g solid solution and the intermetallic compound Cu3Sn takes place: this results in the new phase Cu4Sn.